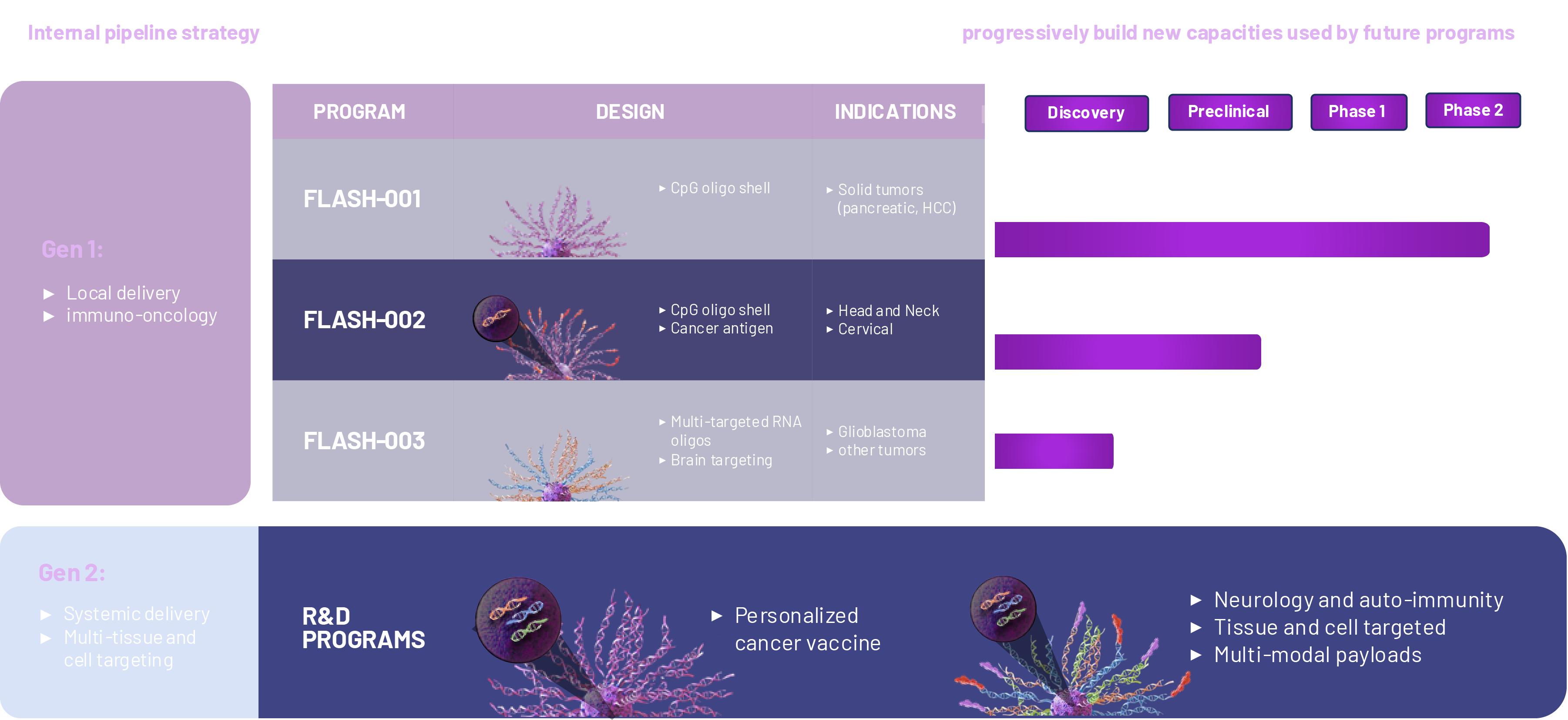
Flashpoint’s pipeline
Flashpoint Therapeutics is a clinical-stage biotechnology company developing a new class of precision-engineered structural nanomedicines. Our platform enables the creation of targeted therapies that co-deliver optimized combinations of therapeutic components directly to individual cells, enhancing drug delivery, stability, potency, and safety. This modular approach supports a broad range of therapies, encompassing modalities like mRNA, DNA, proteins, and CRISPR. With a clinically validated platform demonstrating safety and efficacy, including complete remissions in late-stage cancer, Flashpoint is advancing a robust pipeline of first-in-class nanomedicines through internal development and strategic partnerships.
Our technology
Flashpoint nanostructures are all-in-one formulations that kinetically tune and co-deliver diverse nucleic acid, peptide, and small molecule payloads to the right target cells with precise stoichiometry, optimized structure-function relationships, and synchronized activation kinetics.
The company’s proprietary discovery platform is founded on nanotechnology research developed over the past 10 years in the laboratory of Chad Mirkin, Director of the International Institute for Nanotechnology at Northwestern University.
Validated in numerous in-vivo models, this technology has transformed components that are ineffective in conventional formulations into curative cancer immunotherapies.
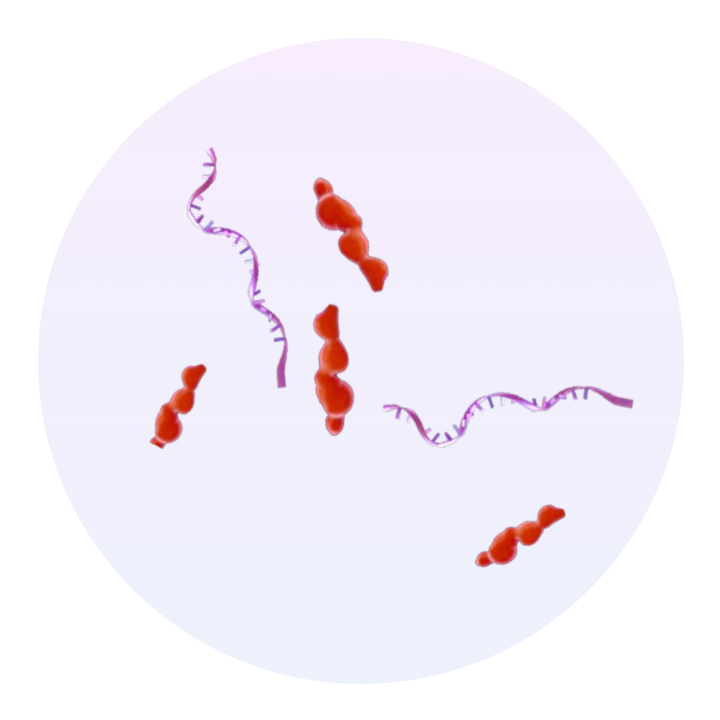
Current Therapies
Unstructured mixture of components
 |
|
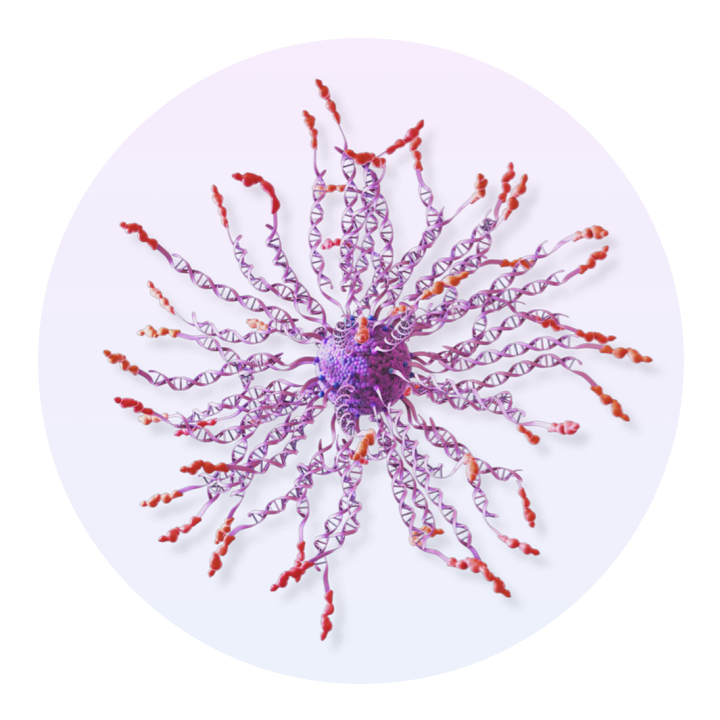
Flashpoint Therapies
Optimized nanoscale architecture
 |
|
Flashpoint’s groundbreaking nanotechnology platform enables the development of therapeutic candidates with superior product profiles by delivering:
- Precise combinations
- To >60 cell types
- With optimized structure-function relationships
- Synchronized multi-pathway activation kinetics
This capability overcomes many major limitations of current therapeutics that have focused on defining potent components but cannot control how they work together.
Flashpoint advantages
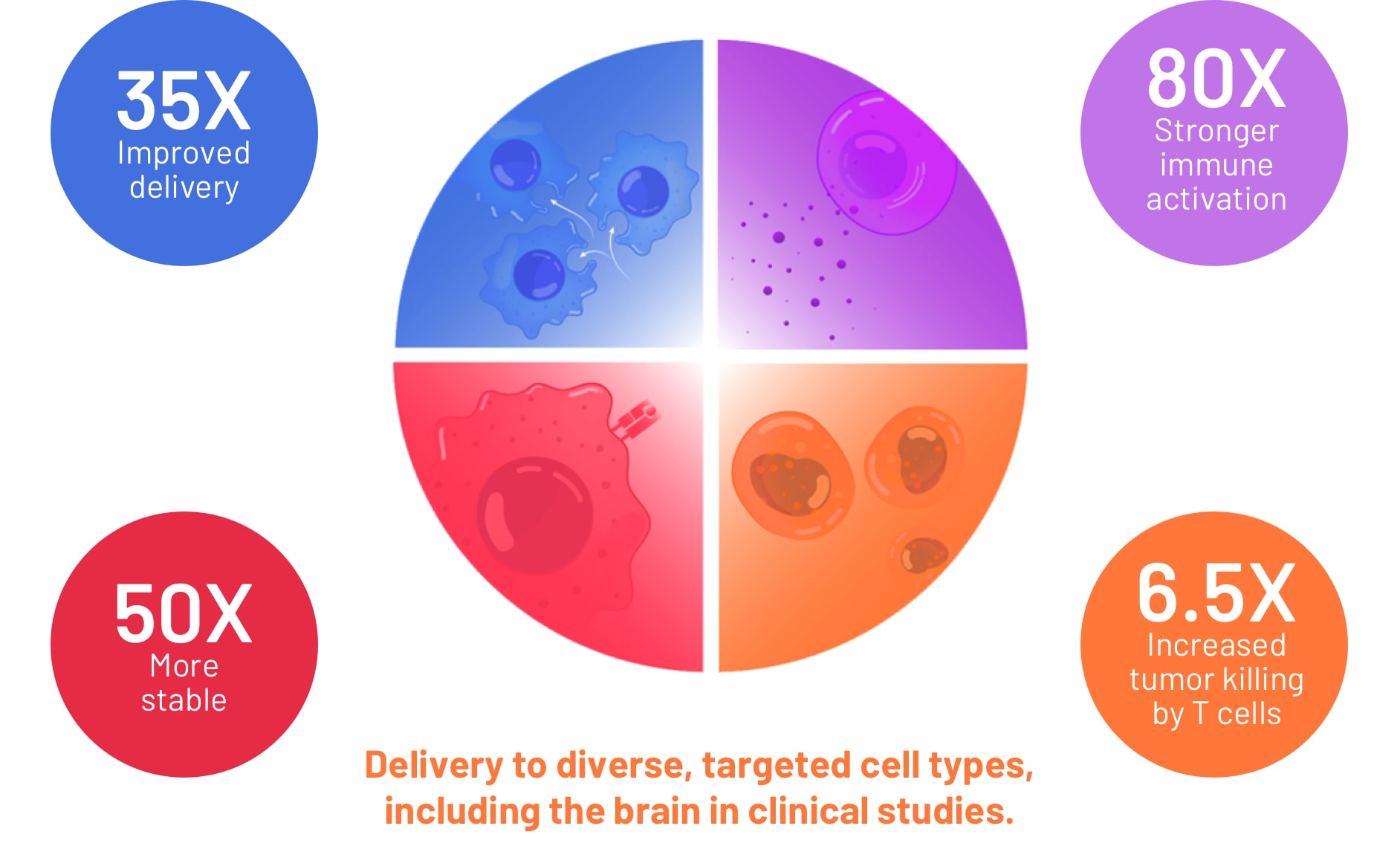
Flashpoint platform has been validated in numerous clinical and animal studies to yield drug candidates with superior product profiles.
Development candidates from the platform have been approved for human clinical testing (IND), manufactured at clinical scale under GMP conditions, and are demonstrated to be highly safe.
Flashpoint Platform
- 650% increased potency over peptide vaccine formulations
- 500% improved potency over LNP encapsulated mRNA
- 250% enhancement of checkpoint inhibitor potency
- 35x enhanced delivery to immune cells
- 80x stronger innate immune activation
- 50x improved stability in circulation
- 30x improved intracellular delivery of peptides
- 44x improved intracellular co-delivery of Cas9 and sgRNAs
- 5x improved gene silencing over lipid encapsulated siRNAs
- Uptake by >60 cell types
- Improved delivery across blood brain barrier in clinical studies
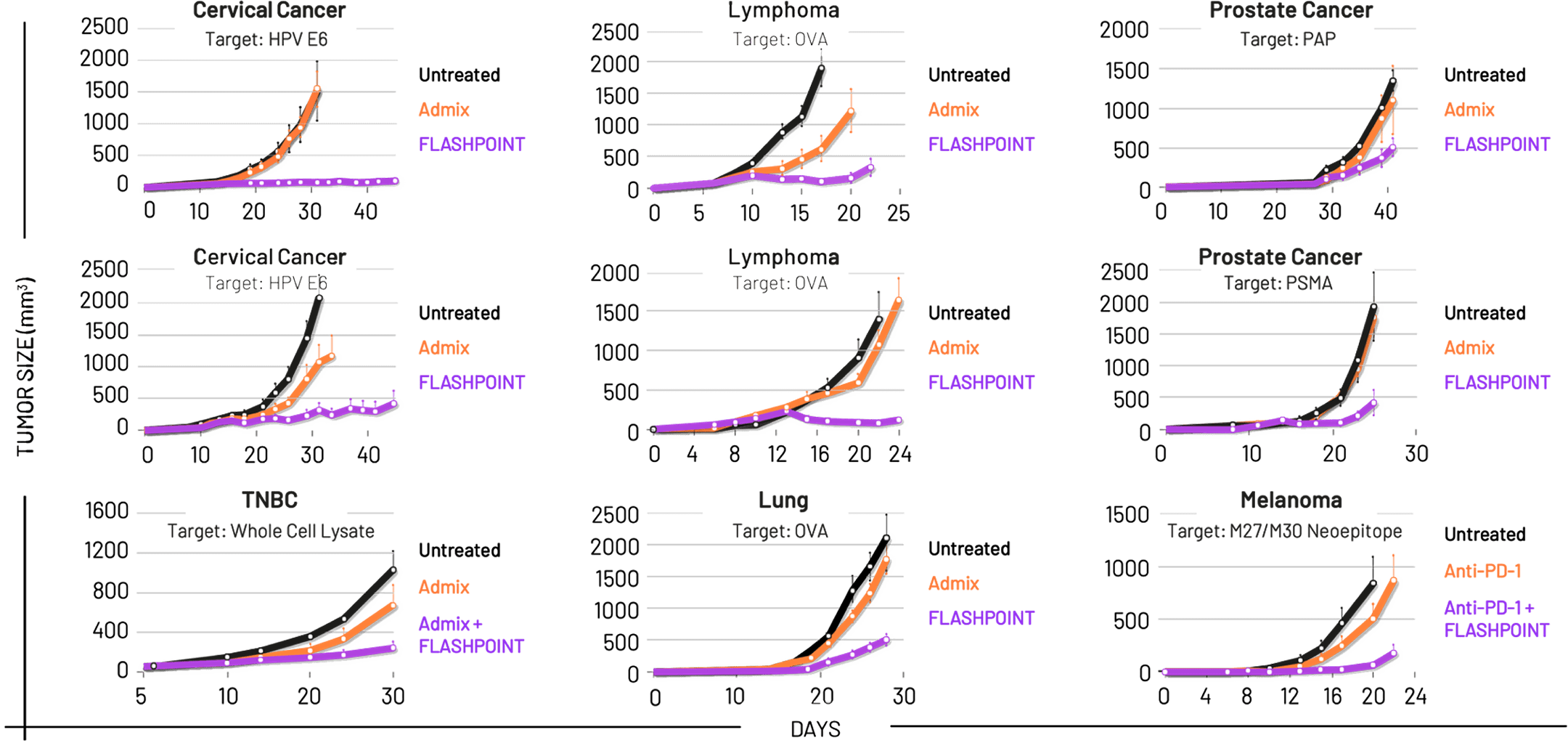
Cancer immunotherapy
Flashpoint is developing first-in-class cancer immunotherapy candidates that delivers precise cocktails of immune stimulatory molecules with nanoscale architecture and activation kinetics optimized to induce robust, coordinated anti-tumor immune response.
Across all 9 in vivo studies performed to date, Flashpoint’s approach has increased the efficacy of immuno-oncology therapeutic components, in some cases leading to curative outcomes.
Next-gen nucleic acid and peptide
nanotherapeutics
Advantages inherited in all formulations |
|
|
|
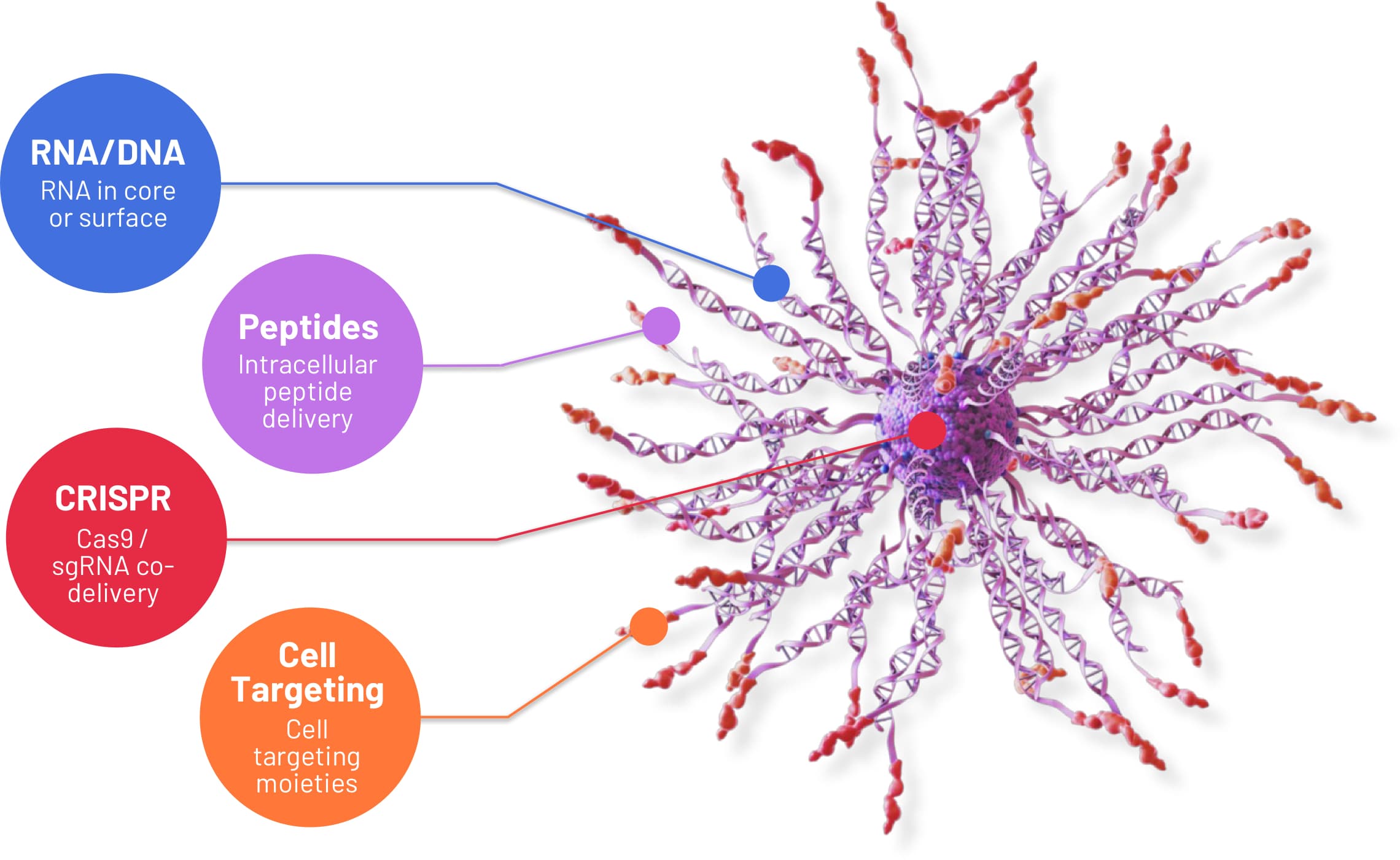
The modular architecture of Flashpoint’s platform enables rapid development of novel candidates delivering combinations of any nucleic acid, peptide, or even small molecular cargo.
In vivo studies have demonstrated the ability to improve targeted delivery, potency, stability, and the safety profile of candidates delivering DNA, RNA, peptides, CRISPR, and cell targeting moieties.
Flashpoint will partner with industry leaders to bring to patients best-in-class multi-targeted RNA and peptide therapeutics.
Our team




















